Keywords to research: Luciferase and Bioluminescent quantum dots
Essential question #1: Non-GMO’s cannot be patented but GMO’s can (synthetic DNA)- who holds the “copyright” when the human genome is manipulated with synthetic gene sequences (e.g., via “vaccination”)? cf.:
Craig Venter: Why I put my name in synthetic genomes.
#2: Nanotechnology/hydrogels in vaccination?
#3: The body as crypto currency coupled to the 5G cloud? patents.google.com/patent/WO2020060606A1/en
#4: Biosensors in Covid vaccine?
Operation Warp Speed is a public–private partnership, initiated by the Trump administration, to facilitate and accelerate the development, manufacturing, and distribution of COVID-19 vaccines, therapeutics, and diagnostics.see Wikipedia
From Wikipedia.com (30.10.2020):
The leader of the Operation Warp Speed project, Moncef Slaoui, had been a board member of the U.S. vaccine developer, Moderna, and divested his shares in Moderna stock, at a potential personal gain of $10 million, raising questions of his neutrality in judging vaccine candidates.[37] Although Slaoui resigned from the Moderna board when named to head Warp Speed, his share value in Moderna stock increased by $3 million in one day when Moderna announced an advance in vaccine clinical research.[37]
Moncef Slaoui is invested in Galvani Bioelectrics, a company concerned with bioelectronic medical applications. At the first C.E.C. Workshop, in Brussels in November 1991, bioelectronics was defined as ‘the use of biological materials and biological architectures for information processing systems and new devices’. N.B. The human nervous system uses electro-chemical signal transduction. Complemetary to classical pharmacological agents based on neurochemistry, bioelectronical applications target the “morse code” of the nervous system, i.e., bioelectronics aims to modulate various levels of neuronal information which is coded in electrical action-potentials. The domain can thus be seen as a counterpart to neurochemistry/neuropharmacology.
Galvani Bioelectronics is a pioneering medical research company dedicated to the development of bioelectronic medicines to treat chronic diseases. Formed through a partnership between two global healthcare companies, GlaxoSmithKline (GSK) and Verily Life Sciences (formerly Google Life Sciences), a subsidiary of Alphabet Inc. Galvani Bioelectronics combines GSK’s life science knowledge with Verily’s expertise in software and electronics for clinical applications.
From Wikipedia.de (30.10.2020):
Die Operation Warp Speed (OWS) ist eine Public-Private-Partnership Initative der Regierung Trump für die Entwicklung und Auslieferung eines SARS-CoV2 Impfstoffes für die Vereinigten Staaten. Präsident Donald Trump teilte im Mitte Mai 2020 mit, dass bis Ende des Jahres 300 Millionen Dosen eines noch nicht existenten Impfstoffes (Stand Oktober 2020) beschafft werden würden.[1][2]
Am 15. Mai 2020 kündigte Präsident Trump die „Operation Warp Speed“ an. Mit ihr sollte die Entwicklung eines Coronavirusimpfstoffes gegen SARS-CoV-2 beschleunigt werden. Geleitet wird diese Operation durch den Pharmamanager Moncef Slaoui und den General der US-Army Gustave Perna.
Die Idee für die Aktion kam Anfang April 2020 auf. Im Juli 2020 waren fünf entwickelte Stoffe in der engeren Auswahl.[2] In der 2. Fernsehdebatte zu US-Wahl 2020 nannte Donald Trump die Operation als Erstes, als er nach seiner Reaktion auf die Pandemie befragt wurde.
- Kathryn Watson, Trump announces top officials for “Operation Warp Speed” for coronavirus vaccine development, CBS News vom 15. Mai 2020.
- Dr Petra Jungmayr: USA: Die fünf aussichtsreichsten COVID-19-Impfstoffkandidaten. 16. Juli 2020, abgerufen am 26. Oktober 2020.
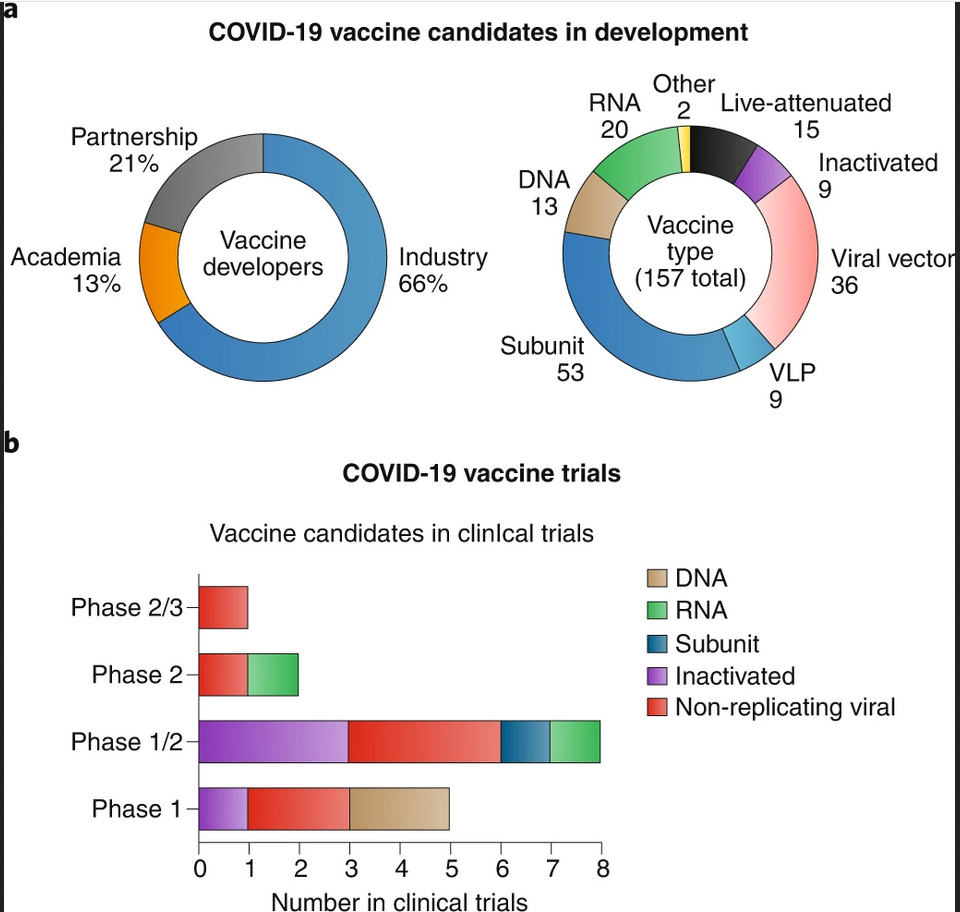
Next-generation vaccines enabled through advances in nanotechnology
Viruses are nanoscale objects and therefore can be regarded as naturally occurring nanomaterials; per that definition, LAVs, IVs and viral vectors are nanotechnologies. Nanoparticles and viruses operate at the same length scale—this is what makes nanotechnology approaches in vaccine development and immunoengineering so powerful. Nanoparticles, natural or synthetic, mimic the structural features of viruses whereas chemical biology, biotechnology and nanochemistry enables the development of next-generation designer vaccine technologies. From a vaccine technology development point of view, this is an exciting time and novel technologies and approaches are poised to make a clinical impact for the first time.
www.nature.com/articles/s41565-020-0737-y
Rice bioengineer reveals dissolving microneedles that also embed fluorescent medical info
The researchers believe the quantum dots are safe to use in this way because they are encapsulated in a biocompatible polymer, but they plan to do further safety studies before testing them in patients.
“Storage, access, and control of medical records is an important topic with many possible approaches,” says Mark Prausnitz, chair of chemical and biomolecular engineering at Georgia Tech, who was not involved in the research. “This study presents a novel approach where the medical record is stored and controlled by the patient within the patient’s skin in a minimally invasive and elegant way.”
The research was funded by the Bill and Melinda Gates Foundation and the Koch Institute Support (core) Grant from the National Cancer Institute.
news.mit.edu/2019/storing-vaccine-history-skin-1218
“Gene-gun”
www.bio-rad.com/en-dk/product/helios-gene-gun-system?ID=42e9d6be-369a-49f8-8fbb-281a0fea6df8
“Gene-gun” usage in plants
www.nature.com/articles/s41565-020-0737-y
The latest report on the worldwide Gene Gun market represents a comprehensive understanding about the futuristic Gene Gun market estimations, based on the historical and current predictions. It is considered to be the significant and professional document that especially focusing on the Gene Gun market drivers, size, leading segments and topological zones. Moreover, the report splits the global Gene Gun market size by volume and value on the basis of product types, applications, well-established vendors and major geographies.
Pivotal industry players involved in this report are:
Bio Rad
Wealtec
DuPont
BTX
Life Technologies
Scientz
Chintek
…
The Global Gene Gun market divided by product types:
Desktop Type
Handhold Type
Gene Gun market segregation by application:
Agriculture
Medical
Other